About Us
As an FDA audited and approved manufacturer it is our responsibility to manufacture products for the public consumer that are of the highest quality. This is a responsibility that we embrace throughout the entire organization. Through our commitment to production technology and quality personnel we are dedicated to manufacturing your product under the strictest of cGMP procedures

Our Facilities
Sheffield Pharmaceuticals operates manufacturing, production and distribution locations in Connecticut, USA.
- Headquartered offices in New London, CT
- 100,000 square foot manufacturing FDA / cGMP facility in New London, CT
- 136,000 square foot Fulfillment and Warehouse Center in Norwich, CT
Sheffield Pharmaceuticals products manufactured and distributed at these locations meet all the requirements of the State of Connecticut Food, Drug, and Cosmetic Act, which conform with the regulations of the FOOD, DRUG, AND COSMETIC ACT of the United States of America.
Sheffield Pharmaceuticals is committed to managing quality activities to ensure that our manufacturing facility operates under cGMP [Current Good Manufacturing Practices], in order to guarantee that the drug products we manufacture have the safety, identity, strength, quality, and purity that it purports or is represented to possess.




Our History

Dr. Washington Wentworth Sheffield was a respected dentist and dental surgeon. In the mid-1870s, he thought of a new tooth cleaning product in a cream form as a replacement for the tooth powders common at the time. He made his own dental cream in his office and added mint extracts to it to improve the flavor He used it on his patients who expressed their liking for it. After establishing his company in 1880, with the help of his son, he constructed a manufacturing plant on his property to produce a mouthwash he had previously invented, as well as the new toothpaste. This manufacturing plant still stands strong and it continues to produce his toothpaste in New London, Connecticut.
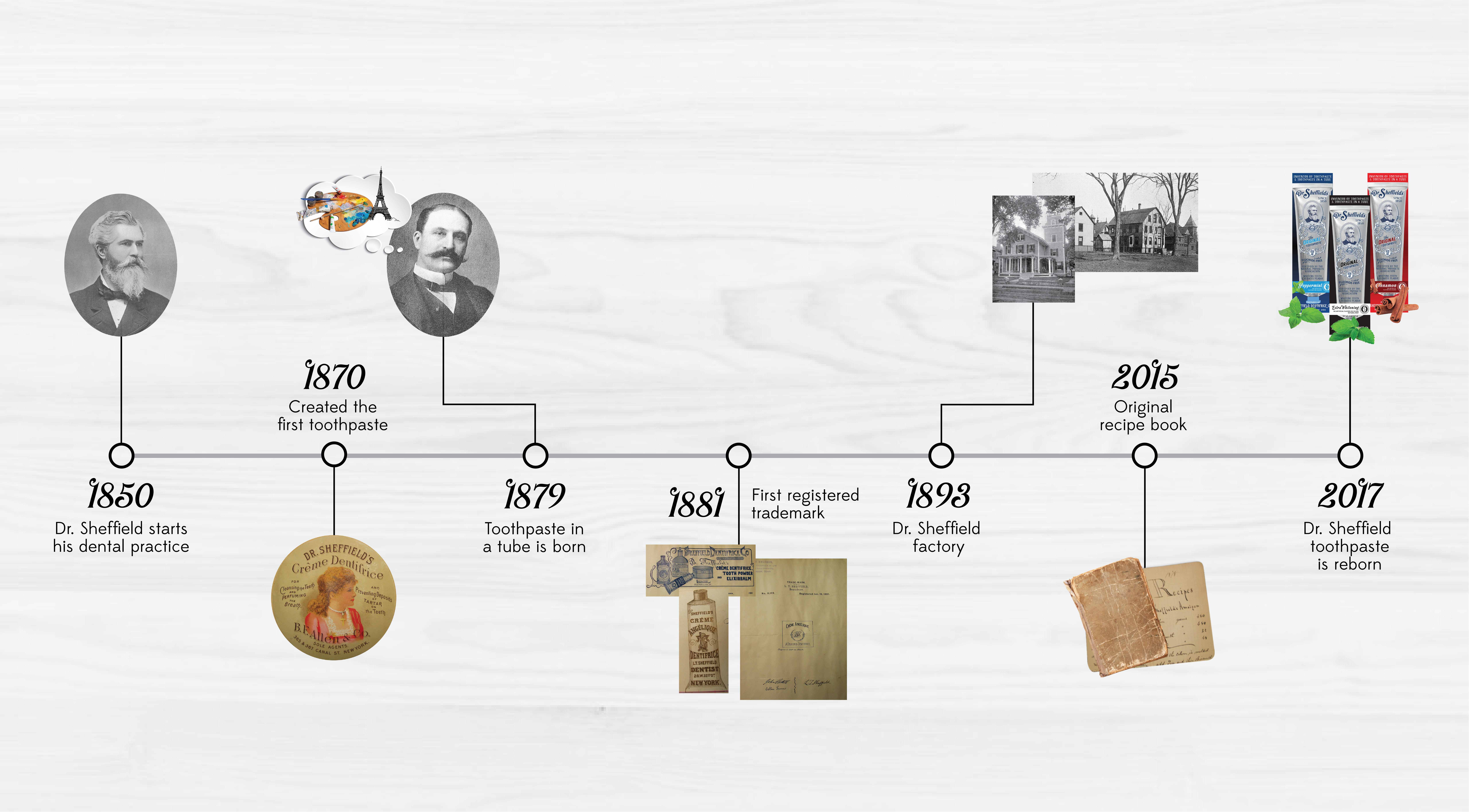
Dr. Lucius T. Sheffield was the only child of Dr. Sheffield. While his father was the first person to invent toothpaste, Lucius was the first person to place the paste into collapsible tubes. Lucius was inspired by Parisian artists preparing their paint palettes. He realized the collapsible tubes they used to squeeze paint onto palettes could be used to squeeze his father’s new toothpaste onto a toothbrush in a sanitary manner.



